Back to 2025 Abstracts
MGMT deficiency is associated with pathological high-risk features of disease in pancreatic neuroendocrine tumors
Leanne Brown
*, Joseph Misdraji, Guannan Gong, Sameer Pandya, Pathik Aravind, Lauren Raymond-King, Theresa Marciano, Joanna Hu, Pamela Kunz, John Kunstman
Yale School of Medicine, New Haven, CT
Background Pancreatic neuroendocrine tumor (PNET) management remains difficult due to rarity and heterogeneity of disease progression. Recently, as therapeutic options have increased, rational sequencing of treatments has become challenging. This can be especially challenging in the adjuvant setting following resection of high-grade PNETs or determining candidacy for metastasectomy. Loss of O-6 methylguanine-DNA methyltransferase (MGMT), a DNA repair enzyme, has been established as a biomarker for PNET susceptibility to alkylating agents but the relationship of MGMT deficiency with clinicopathologic variables and survivorship is unknown. This study aims to characterize PNET MGMT expression to further explore its role as a novel biomarker to direct PNET treatment sequencing.
Study Design A single institution retrospective review of patients undergoing resection between 1994-2022 for well differentiated PNET was performed. MGMT expression within primary tumors was profiled by immunohistochemistry (IHC) into three groups, absent, diminished, and intact expression. Clinical information obtained via chart review included demographics, surgical pathology, and follow up. Demographic categorical variables were assessed via Chi-squared and Kruskall-Wallis test, respectively. Ordinal logistic and linear regression models were built to investigate the association of the remaining categorical variables and tumor size with MGMT expression, respectively. For patients who were completely resected at time of index surgery, Recurrence-Free Survival (RFS) as defined by radiographic evidence of recurrence and Overall Survival (OS) were analyzed via the Kaplan-Meier method.
Results A total of 129 well differentiated PNETs were identified, of which 41 (31.8%), 38 (29.5%), and 50 (38.8%) represent MGMT-absent, MGMT-diminished, and MGMT-intact tumors, respectively. Between MGMT groups, there was no difference in age, sex, race, or tumor location. Decreased (diminished or absent) MGMT expression was associated with larger median tumor size (R
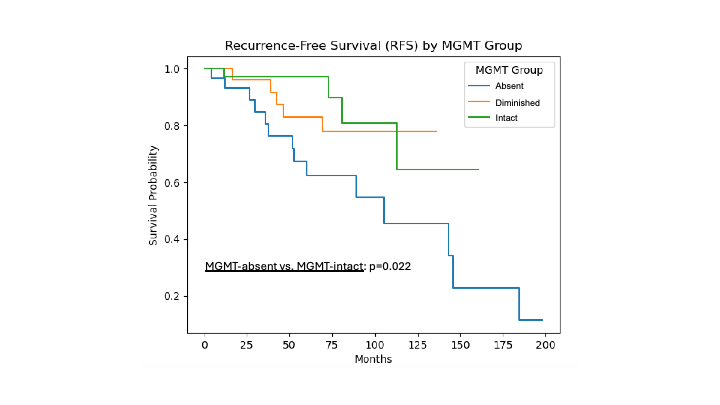
2=0.110, p<0.001) and higher tumor grade (OR 0.54 [95% CI 0.34-0.87], p=0.011). Decreased MGMT expression was also associated with higher likelihood of lymphovascular invasion and regional nodal metastasis (OR 0.44 [95% CI 0.28-0.70], p = 0.001; OR 0.43, [95% CI 0.26-0.70], p=0.001, respectively). Patients with MGMT-absent tumors exhibited a significantly worse RFS with a median survival of 105.3 months compared to intact expression in which the median survival was not reached (p=0.022). There was no significant difference in OS between the MGMT groups.
ConclusionsDiminished or absent MGMT expression is frequent in PNET and was associated with high-risk pathological features including larger tumor size, higher grade, nodal metastases, and earlier recurrence. MGMT profiling enables identification of aggressive PNETs and merits prospective investigation as a promising novel biomarker to guide treatment selection in these patients.