Characterization of Peripheral Blood T-Lymphocyte Populations Following Intratumoral Anti-PD-1 Injection in a Humanized Mouse Model
Ryan J. Hendrix, *Kurt Schultz, *Michael Brehm, Giles F. Whalen
University of Massachusetts Medical School, Worcester, MA
OBJECTIVE: We aim to characterize the changes in peripheral blood following intratumoral (IT) anti-PD-1 injection and assess its efficacy in stimulating specific T-Lymphocyte (TL) populations compared to intravenous (IV) anti-PD-1 injection.
DESIGN: Randomized controlled trial.
SETTING: Biosafety Level 2 (BSL-2) laboratory and animal facilities at an academic medical center.
INTERVENTION: 34 NOD-scid IL2rgnull (NSG) mice engrafted with human hematopoietic stem cells (HSCs) had patient derived xenograft (PDX) melanoma tumors implanted subcutaneously. Once tumor volumes of 100mm3 were achieved, mice were randomized to 1 of 3 study arms involving a single injection: IV anti-PD-1 at 10mg/kg (n=13), IT anti-PD-1 at 20mg/kg (n=13), or IT PBS solution (n=8). Mice were sacrificed on days 6 and 12 post injection.
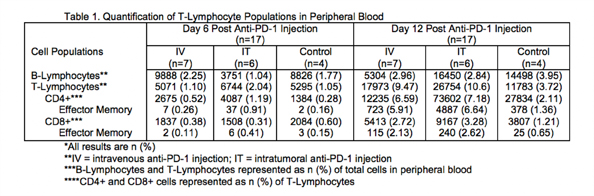
MAIN OUTCOME MEASURE: Peripheral blood, spleen, tumor, liver, kidney, and lung tissue specimens were obtained. Cell counts and proportions were reported for CD8+, CD4+, and effector memory T-Lymphocyte populations. Ten-color flow cytometry panels were used to identify these specific subpopulations.
RESULTS: Proportion of TLs in peripheral blood was greatest in the IT group at days 6 (2.04% vs 1.10%) and 12 (10.6% vs 9.47%). Similar trends were observed for all TL subpopulations (Table 1). Expression of effector memory TLs increased from days 6 to 12 but was most robust following IT injection.
CONCLUSION: IT injection of anti-PD-1 appeared to produce a different profile of lymphocyte effector memory cells than IV injection of anti-PD-1. Further investigation of the implications of these findings and the potential of immune checkpoint inhibitors as neoadjuvant cancer therapy is warranted.